Cardiac rhythmic activity is generated by "pacemaker" cells, which in mammals are located in the sino-atrial node (SAN). Action potentials of SAN cells have a special phase, called diastolic (or pacemaker) depolarization: at the end of an action potential, the pacemaker depolarization slowly takes the membrane voltage up to threshold for firing of a new action potential, thus generating repetitive activity. What is the mechanism underlying the pacemaker depolarization?
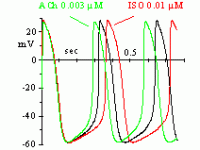
In 1979, Brown, DiFrancesco & Noble described a "funny" (If) current, so called because of its unusual properties, in cardiac pacemaker cells. Since If was inward and slowly activating on hyperpolarization at voltages in the diastolic range, the authors proposed that it was involved in the generation of spontaneous activity; the current was also increased by adrenaline, indicating an involvement in the catecholamine-induced acceleration of pacemaker activity and hence of heart rate. In another cardiac preparation displaying spontaneous activity, the Purkinje fibre, a "pacemaker" current had been observed previously, but it had been described by as a pure K+ current, outward at diastolic voltages (IK2 current, Noble & Tsien, 1968). The Purkinje fibre's IK2 current was similar to the nodal If in several aspects, and the apparently different ionic nature of the two currents remained a puzzling issue until in 1981, a reinterpretation of the IK2 current was proposed (DiFrancesco 1981a,b) demonstrating that the two currents were indeed identical, and IK2 had been erroneously intrepreted for over 10 years as a pure K+ current. This demonstrated the identity of pacemaker components in different cardiac cells. Following its early description, If was extensively investigated in cardiac cells (DiFrancesco, 1993). If is a mixed Na+ and K+ current, activated on hyperpolarization from a threshold of about -40/-50 mV. When at the end of an action potential the membrane repolarizes below this threshold, If is activated and supplies inward current, which is responsible for generation of the diastolic depolarization phase. If is also modulated by intracellular cAMP, by an action exerted directly on f-channels and not mediated by phosphorylation (DiFrancesco & Tortora, 1991). Raising cAMP shifts the If activation curve to more positive voltages, thus providing more inward current for the pacemaker depolarization. This action mediates the modulation of cardiac rate by the autonomic nervous system. In cardiac cells, cAMP synthesis is stimulated and inhibited by ß-agonists and muscarinic agonists, respectively; thus, sympathetic stimulation accelerates, and vagal stimulation slows, cardiac rate by increasing and decreasing, respectively, If at diastolic potentials via changes in cAMP level. Moderate vagal activity, such as the one associated to basal vagal tone, controls cardiac rate by modulation of If, rather than by the opening of ACh-activated K+ channels as was previously thought (DiFrancesco, Ducouret & Robinson, 1989). If has a fairly low single-channel conductance (about 1 pS, DiFrancesco, 1986). Binding of cAMP molecules to f-channels induces a conformational change of the protein, associated with an increased probability of channel opening upon hyperpolarization (DiFrancesco & Mangoni, 1994). This mechanism can be interpreted in terms of an allosteric cAMP-induced channel activation whereby cAMP binds preferentially, and thus stabilizes, the "open" configuration of the channel (DiFrancesco, 1999). If-like currents have been described in a variety of neuronal preparations (Ih current, Pape, 1996). Generally, the role of Ih in neurons is based on its ability to generate a depolarization following either membrane hyperpolarization, or a receptor- mediated modification of intracellular cAMP. Thus, Ih can contribute to control of resting potential and modulation of excitability, or to firing discharge. In sensory neurons Ih is involved in the perception of external stimuli, or in modulating the transduction of sensory stimuli into electrical signalling. Ih is also expressed in some pre-synaptic membranes, where it is involved in plasticity phenomena (Beaumont & Zucker, 2000; Mellor et al., 2002). The relevance of f-channels to generation and control of heart rate makes them important pharmacological targets. Several heart rate-reducing agents-molecules able to induce bradycardia without inotropic side effects- have been developed, such as UL-FS 49, ZD 7288 and ivabradine. Specific heart-rate reducing agents have a potential for therapeutic use in all those cases where it is useful to slow heart rate without altering other cardiovascular functions. Substantial advancement in the knowledge of the molecular properties of pacemaker channels has been obtained with the cloning in the late ´90s of the HCN family of channels (Hyperpolarization-activated, Cyclic-Nucleotide-gated). HCN channels are characterized by a structure similar to that of K+ voltage-dependent (Kv) and cyclic-nucleotide gated (CNG) channels. The properties of HCN channels expressed heterologously show that they are the molecular determinants of native pacemaker channels in the heart and nervous system.